Comprehensive In-vitro Proarrhythmia Assay, CiPA
-
hiPSC
2 weeks

-
Cardiomyocytes


-
Cardiac safety service
Research & Development
The Comprehensive in Vitro Proarrhythmia Assay (CiPA)
initiative was established to develop a new paradigm for
assessing proarrhythmic risk, building on the emergence of
new technologies and an expanded understanding of
torsadogenic mechanisms beyond hERG block.
T&R Biofab is actively supporting the CiPA initiative to improve
the current regulatory guidance for preclinical cardiac safety
assessment. We are conducting safety verification of candidate drugs according to changes in international and domestic guidelines (ICH S7B E14). Our research on cardiotoxicity evaluation in various disease patient models are published in various peer-reviewed scientific journals.
-
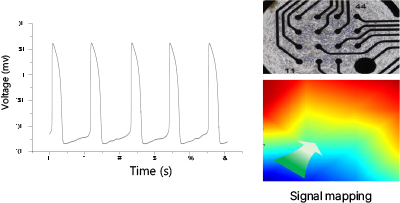
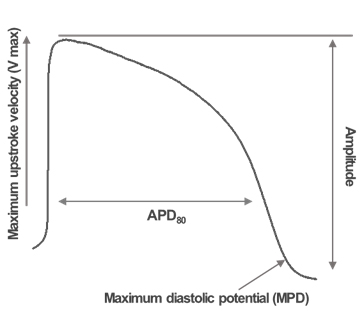
Action Potential Duration
-
Drug "A" (T&R hCardiomyocytes, 30 min)
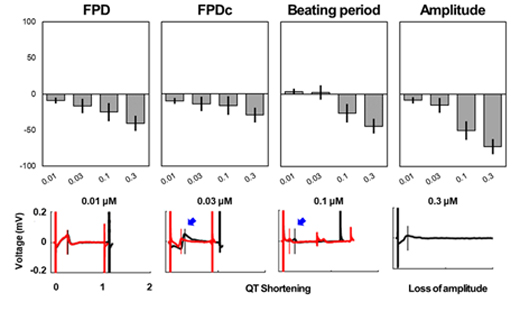
-
Drug "B" (T&R hCardiomyocytes, 30 min)
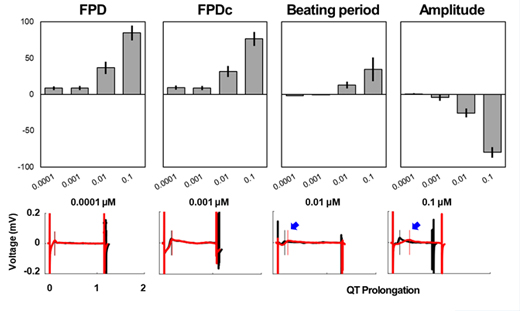
Cardiotoxicity Assessment Service
01Cardiotoxicity evaluation using pre-defined drug concentration and conditions requested by the customer
02Patient-specific toxicity evaluation using cardiomyocytes of the same sex and similar age
03Various selection for analysis (6 to 96 wells)
Related articles
Antiviral activity and safety of “Remdesivir” against SARS-CoV-2 (COVID-19) infection in human pluripotent stem cell-derived cardiomyocytes. Antiviral Research, 2020